COVID-19 has changed the world. After it rapidly spread to every continent, it became a worldwide emergency. Its causative agent, severe acute respiratory syndrome coronavirus 2 or SARS-CoV-2, has infected millions of individuals. These individuals can vary from asymptomatic to mild and moderate symptoms [1].
As scientists are still conducting studies on the immune impact of this virus, I explored the literature to identify research conducted on the virus immune impact and vaccine against SARS-CoV-2.
On July 28th, as countries began to ease their regulations and follow health expert guidelines, the CDC recorded 4,225,687 COVID-19 cases in the US and 146,546 deaths [2].
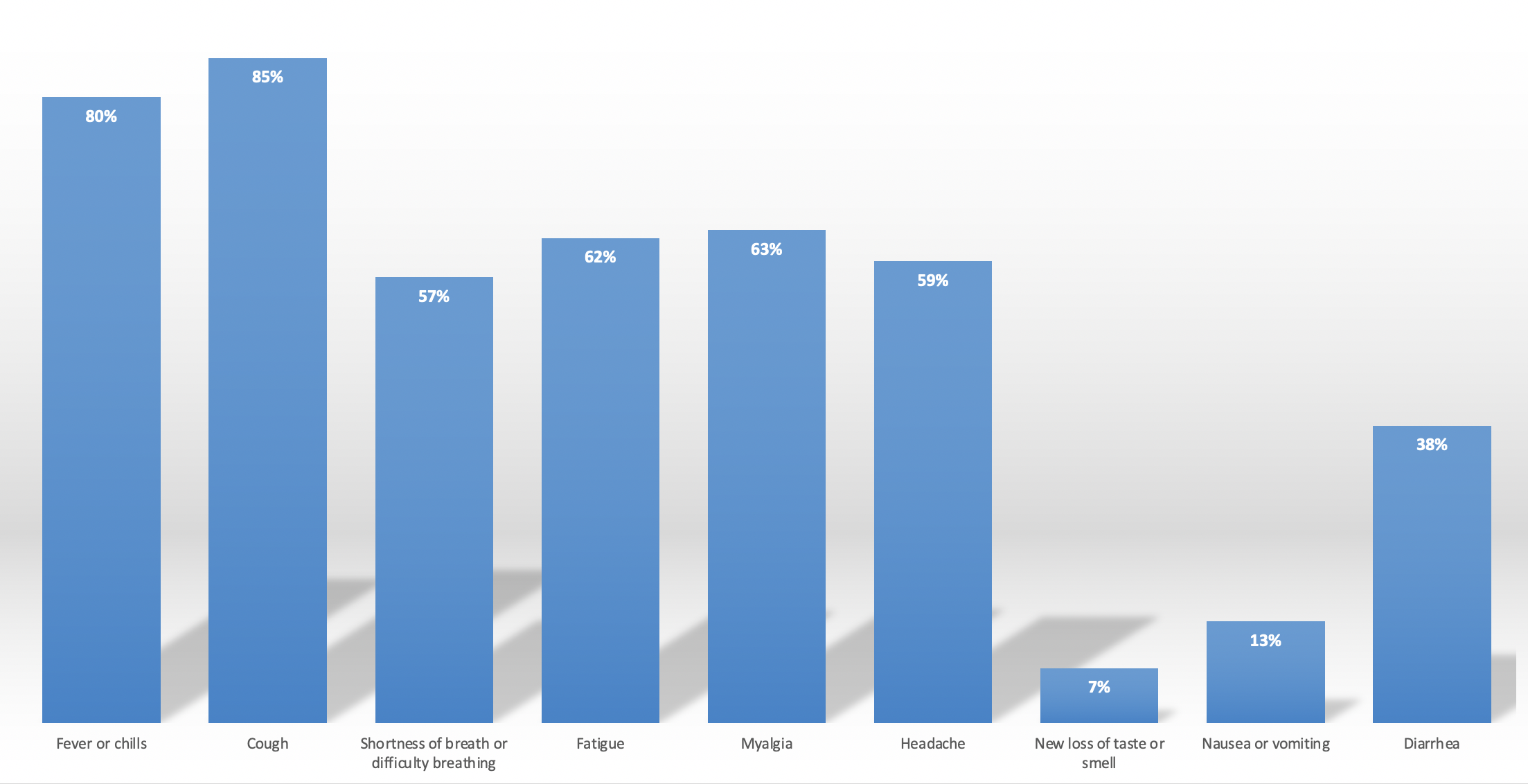
Since December 2019, when COVID-19 has first been made award in China, it spread and has been handled in different ways around the world which can be used to create a timeline [3]. Even though each infected individual showed a variety of symptoms, the most common symptoms have been identified and reported on a graph [4].
Chart 1 showcases a COVID-19 pandemic development timeline [3,5].
 Download this Timeline
Download this TimelineChart 2 showcases the prevalence of Covid-19 Symptoms among hospitalized patients, with coughing and fever being most common symptoms observed in patients [4].
SARS-CoV-2 causes patients to have abnormalities of granulocytes and monocytes, lymphopenia, increased antibodies, cytokines production, lymphocyte activation, and dysfunction [6,7,8].
Chart 3 showcases the immune pattern of COVID-19 credit Yang et al. [9]

In the case of patients who tested positive after recovering from the infection, experts believe it could be false positive or tests detected remnants of previous infection. Otherwise patients may just have not fully recovered; further research on the matter is needed [10].
One promising vaccine against COVID-19 that is currently in phase clinical trial is the mRNA-1273. A mRNA vaccine, mRNA-1273, is more precisely a novel lipid nanoparticle (LNP)-encapsulated mRNA-based vaccine, that encodes for prefusion stabilized spike (S) protein of SARS-CoV-2. Phase 1 of the vaccine was to assess the safety through a dose-ranging clinical trial done on 120 participants both males and non-pregnant females 18 years or older, and based on the serum-neutralizing activity and antibody level, the vaccine was able to move to a second phase. Scientists during phase 2 of the clinical trial were assessing the reactogenicity, immunogenicity, and safety of two vaccinations of mRNA-1273 given 28 days apart. Phase 3 protocol of the vaccine was approved and in July the clinical trial began [10, 11, 12].
Click below to read more about Immunological Response to COVID-19 Infection and Vaccine Development. You can also download our report in the link below.

Bachelor of Science in Cell and Molecular Biology | University of South Florida College of Arts and Sciences
Masters of Public Health Candidate: Global Communicable Disease | University of South Florida College of Public Health
(1) Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. The Lancet. 2020;395(10223):497-506. doi:10.1016/S0140-6736(20)30183-5
(2) CDC COVID Data Tracker. Accessed July 28, 2020. https://www.cdc.gov/covid-data-tracker/#cases
(3) World Health Organization. Timeline of WHO’s response to COVID-19. Published June 30, 2020. Accessed July 27, 2020. https://www.who.int/news-room/detail/29-06-2020-covidtimeline
(4) Burke RM. Symptom Profiles of a Convenience Sample of Patients with COVID-19 — United States, January–April 2020. MMWR Morb Mortal Wkly Rep. 2020;69. doi:10.15585/mmwr.mm6928a2
(5) Machhi J, Herskovitz J, Senan AM, et al. The Natural History, Pathobiology, and Clinical Manifestations of SARS-CoV-2 Infections. J Neuroimmune Pharmacol. Published online July 21, 2020:1-28. doi:10.1007/s11481-020-09944-5
(6) Zhang B, Zhou X, Zhu C, et al. Immune phenotyping based on neutrophil-to-lymphocyte ratio and IgG predicts disease severity and outcome for patients with COVID-19. medRxiv. Published online March 16, 2020:2020.03.12.20035048. doi:10.1101/2020.03.12.20035048
(7) Liu Y, Sun W, Li J, et al. Clinical features and progression of acute respiratory distress syndrome in coronavirus disease 2019. medRxiv. Published online February 27, 2020:2020.02.17.20024166. doi:10.1101/2020.02.17.20024166
(8) Liu J, Li S, Liu J, et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55:102763. doi:10.1016/j.ebiom.2020.102763
(9) Yang L, Liu S, Liu J, et al. COVID-19: immunopathogenesis and Immunotherapeutics. Signal Transduct Target Ther. 2020;5(1):1-8. doi:10.1038/s41392-020-00243-2
(10) Kirkcaldy RD, King BA, Brooks JT. COVID-19 and Postinfection Immunity: Limited Evidence, Many Remaining Questions. JAMA. 2020;323(22):2245-2246. doi:10.1001/jama.2020.7869
(11) Phase 3 clinical trial of investigational vaccine for COVID-19 begins. National Institutes of Health (NIH). Published July 25, 2020. Accessed July 28, 2020. https://www.nih.gov/news-events/news-releases/phase-3-clinical-trial-investigational-vaccine-covid-19-begins
(12) National Institute of Allergy and Infectious Diseases (NIAID). Phase I, Open-Label, Dose-Ranging Study of the Safety and Immunogenicity of 2019-NCoV Vaccine (MRNA-1273) in Healthy Adults. clinicaltrials.gov; 2020. Accessed July 28, 2020. https://clinicaltrials.gov/ct2/show/NCT04283461
(13) Jackson LA, Anderson EJ, Rouphael NG, et al. An mRNA Vaccine against SARS-CoV-2 — Preliminary Report. N Engl J Med. 2020;0(0):null. doi:10.1056/NEJMoa2022483